Basic Introduction To Collision Theory
Collision theory is used to explain how a chemical reaction occurs and what happens (at a particle-level) when a chemical reaction takes place.
The collision theory states that: (Important!!)
- For a reaction to take place, the reactant particles HAVE to collide.
- Not all collisions will result in a chemical reaction (i.e result in the formation of products).
- Collisions that result in a chemical reaction are termed as effective collisions.
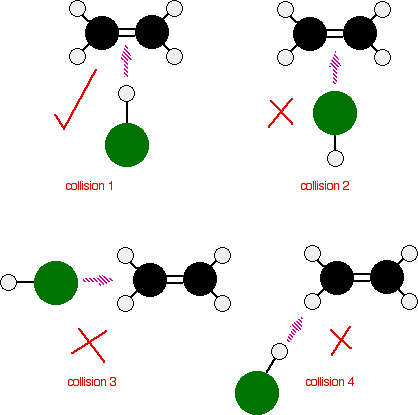
- An effective collision can only occurs when the reactant particles:
- have more than or equal to the activation energy, $\text{E}_{a}$, of the reaction; AND (Refer to subsequent paragraphs for explanation on activation energy)
- collide at the correct/proper orientation. (refer to figure below)
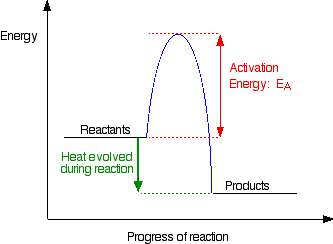
Activation Energy
The activation energy, $\text{E}_{a}$ of a chemical reaction is the minimum amount of kinetic energy that the reactant particles need in order to have an effective collision.
The activation energy of a reaction can be shown on the energy profile for a reaction. The figure above shows the energy profile of a simple exothermic reaction.
- The activation energy is shown as the energy barrier which the reactant particles need in order to start the reaction.
- If the reactants do not have enough energy to overcome the energy barrier, there will be no effective collision and the reaction will not occur.
We know that the rate of reaction depends on the number of effective collision between reacting particles:
The greater the number of effective collisions per unit time $\rightarrow$ the higher the rate of reaction.
- The higher the number of collisions per unit time, the higher the rate of reaction.
- The higher the amount of kinetic energy possessed by the reactant particles, the higher the rate of reaction.
Hence, there are a few factors which affect the rate of a reaction:
- Concentration
- Pressure
- Particle size
- Temperature
- Presence of catalyst
Notice that the listed factors affect (directly or indirectly) the number of collisions per unit time or the number of reactant particles having energy equal to or more than the activation energy. (We will explore these factors in the next post.)