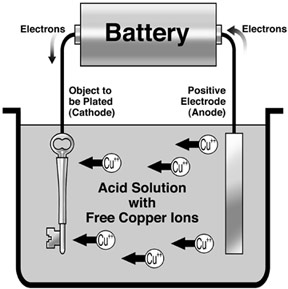
Electroplating
Electroplating is used to coat one metal with another metal by using electrolysis. Electroplating is usually done to improve the appearance of the metal or prevent the corrosion of the metal.
Chromium plating is used to prevent iron from rusting.
The cathode is the object to be plated, while anode is the desired metal to coat the object. The electrolyte solution must contain ions of the same metal for plating. During electrolysis, the anode will dissolve into the solution. The ions produced will migrate to the cathode where they are discharged and deposited as a layer on the cathode.
It is important to ensure that the cathode is electrically conductive. (If not, the electrolysis does not work.)
Case Study: Silver Plating
Electrodes: Silver anode, Cathode is object to be plated with silver
Electrolyte: Solution of soluble silver salt. (E.g. Silver nitrate, $AgNO_{3} (aq)$)
$$AgNO_{3} (aq) \rightarrow Ag^{+} (aq) + NO_{3}^{-} (aq)$$
$$H_{2}O (l) \rightleftharpoons H^{+} (aq) + OH^{-} (aq)$$
Ions present in solution: $Ag^{+}$, $H^{+}$, $NO_{3}^{-}$, $OH^{-}$
Reaction at CATHODE:
- $Ag^{+}$ and $H^{+}$ is attracted to the cathode.
- $Ag^{+}$ is preferentially discharged.
- Silver metal is deposited on the cathode.
- $Ag^{+} (aq) + e^{-} \rightarrow Ag (s)$
Reaction at ANODE:
- $NO_{3}^{-}$ and $OH^{-}$ is attracted to the anode.
- Neither $NO_{3}^{-}$ nor $OH^{-}$ is discharged.
- Each silver atom loses one electron to form one $Ag^{+}$ ion.
- The silver anode dissolves into the solution.
- $Ag (s) \rightarrow Ag^{+} (aq) + e^{-}$
Extraction Of Reactive Metals
Reactive metals are the metals that occupy the top positions in the electrochemical series. Metals that are higher than zinc in the electrochemical series are extracted using electrolysis. These very reactive metals cannot be extracted by other metals such as reduction with carbon. The reactive metals are obtained by electrolyzing a molten ionic compound of metal.
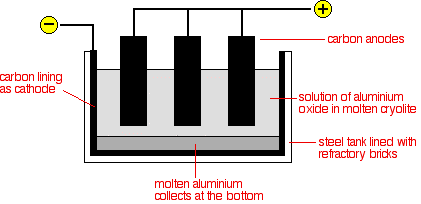
Case Study: Extraction Of Aluminium
Aluminium is extracted from aluminium oxide ($Al_{2}O_{3}$) or known as bauxite.
The electrolytic cell is an iron tank lined with carbon, which acts as the cathode. The anodes are blocks of carbon dipped into the electrolyte. The electrolyte is a solution of molten aluminum oxide in molten cryolite. Cryolite acts as a solvent to dissolve aluminium oxide and as an impurity to lower the melting point of aluminium oxide. The electrolytic cell is maintained at around $900^{\circ}\text{C}$.
Electrodes: Carbon
Ions present in electrolyte: $Al^{3+}$, $O^{2-}$
Reaction at cathode: $Al^{3+} (l) + 3 e^{-} \rightarrow Al (l)$
Reaction at anode: $2 O^{2-} (l) \rightarrow O_{2} (g) + 4 e^{-}$
Aluminium ions are discharged at the cathode, forming a pool of molten aluminium at the bottom of the tank.
At high temperature, oxygen reacts with the carbon anode to form carbon dioxide gas. Hence, the anodes are slowly burnt away as carbon dioxide gas and needs to be replaced frequently.