Aqueous compounds are a mixtures of two electrolytes – the compound and water.
- Water is a weak electrolyte: It ionizes a little to give $H^{+}$ ions and $OH^{-}$ ions: $H_{2}O (l) \rightleftharpoons H^{+} (aq) + OH^{-}(aq)$
- When there is more than one type of anions and cation present, preferential discharge will be carried out. (More about this later) The selection of ions for preferential discharge will be dependent on the position of the ions in the reactivity/electrochemical series, the concentration of ions and the types of electrodes.
Reactivity/Electrochemical Series
| Cation | Anion |
|---|---|
| $K^{+}$ | $F^{-}$ |
| $Ca^{2+}$ | $SO_{4}^{2-}$ |
| $Na^{+}$ | $NO_{3}^{-}$ |
| $Mg^{2+}$ | $Cl^{-}$ |
| $Al^{3+}$ | $Br^{-}$ |
| $Zn^{2+}$ | $I^{-}$ |
| $Fe^{2+}$ | $OH^{-}$ |
| $Sn^{2+}$ | |
| $Pb^{2+}$ | |
| $H^{+}$ | |
| $Cu^{2+}$ | |
The ion lower down in the electrochemical series will show a greater tendency to be deposited than another which is higher up.
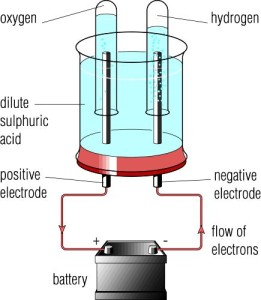
Case Study: Electrolysis Of Dilute Sulphuric Acid $H_{2}SO_{4} (aq)$
Electrodes: Carbon rods
Electrolyte: Dilute sulphuric acid
$$H_{2}SO_{4} (aq) \rightarrow 2H^{+} (aq) + SO_{4}^{2-} (aq)$$
$$H_{2}O (l) \rightleftharpoons H^{+} (aq) + OH^{-} (aq)$$
Ions present in electrolyte: $H^{+}$, $OH^{-}$ and $SO_{4}^{2-}$
Reaction at CATHODE:
- $H^{+}$ ions are attracted to the cathode
- Each $H^{+}$ ions gains one electron from the cathode to form one hydrogen atom.
- Effervescence of colourless, odourless hydrogen gas is seen at the cathode.
- $2H^{+} (aq) + 2e^{-} \rightarrow H_{2}(g)$
Reaction at ANODE:
- $OH^{-}$ and $SO_{4}^{2-}$ ions are attracted to the anode.
- $OH^{-}$ is lower than $SO_{4}^{2-}$ in the electrochemical series.
- $OH^{-}$ ion is preferentially discharged.
- Each $OH^{-}$ ion loses one electron to the anode to form oxygen gas and water molecules.
- Effervescence of colourless and odourless oxygen gas is seen at the anode.
- $4OH^{-} (aq) \rightarrow O_{2} (g) + 2H_{2}O (l) + 4 e^{-}$
Overall reaction:
- Water is decomposed into hydrogen and oxygen gas in the volume ratio of 2:1.
- $2H_{2}O (l) \rightarrow 2H_{2} (g) + O_{2} (g)$
- $H_{2}SO_{4}$ is NOT decomposed by electricity. $H_{2}SO_{4}$ is added to increase the electrical conductivity of the electrolyte.
- As the water is decomposed, the concentration of sulphuric acid increases.
Case Study: Electrolysis Of Aqueous Sodium Hydroxide $NaOH (aq)$
Electrode: Carbon Rods
Electrolyte: Aqueous sodium hydroxide
$$NaOH (aq) \rightarrow Na^{+} (aq) + OH^{-} (aq)$$
$$H_{2}O (l) \rightleftharpoons H^{+} (aq) + OH^{-} (aq)$$
Ions present in the electrolyte: $H^{+}$, $OH^{-}$ and $Na^{+}$ ions
Reaction at CATHODE:
- $H^{+}$ and $Na^{+}$ ions are attracted to the cathode.
- $H^{+}$ is lower than $Na^{+}$ in the electrochemical series.
- $H^{+}$ ion is preferentially discharged.
- Each $H^{+}$ ion gain one electron from the cathode to form one hydrogen atom.
- Effervescence of colourless, odourless hydrogen gas is seen at the cathode.
- $2H^{+} (aq) + 2e^{-} \rightarrow H_{2} (g)$
Reaction at ANODE:
- $OH^{-}$ ions are attracted to the anode.
- Each $OH^{-}$ ion loses one electron to the anode to form oxygen gas and water molecules.
- Effervescence of colourless and odourless oxygen gas is seen at the anode.
- $4OH^{-} (aq) \rightarrow O_{2} (g) + 2H_{2}O (l) + 4e^{-}$
Overall Reaction:
- Water is decomposed into hydrogen and oxygen gas in the volume ratio of 2:1.
- $2H_{2}O (l) \rightarrow 2H_{2} (g) + O_{2} (g)$
- $NaOH$ is not decomposed.
- As water is decomposed, the concentration of $NaOH$ increases.
Concentration Of Ions
An ion that is higher in the electrochemical series could get preferentially discharged over an ion that is lower in the series IF the concentration of ion (higher in electrochemical series) is much higher than the other ion (lower in electrochemical series).
- One example: In a solution of concentrated hydrochloric acid, chlorine gas will be formed at the anode. (More about this later) Recall that if a solution of dilute hydrochloric acid is used, oxygen gas will be formed at the anode.
However, there are some ions which ‘like’ to be in the ion-state too much to be discharged due to this effect. The ions are:
- Cations: $K^{+}$, $Na^{+}$, $Ca^{2+}$, $Mg^{2+}$ and $Al^{3+}$
- Anions: $F^{-}$, $SO_{4}^{2-}$, $NO_{3}^{-}$ and $CO_{3}^{2-}$
These special ions are NOT discharged even if they are present in higher concentration than other ions.
Case Study: Electrolysis Of Concentrated Hydrochloric Acid $HCl (aq)$
Electrodes: Carbon rods
Electrolytes: Concentrated hydrochloric acid
$$HCl (aq) \rightarrow H^{+} (aq) + Cl^{-} (aq)$$
$$H_{2}O(l) \rightleftharpoons H^{+} (aq) + OH^{-} (aq)$$
Ions present in electrolyte: $H^{+}$, $OH^{-}$ and $Cl^{-}$
Reaction at CATHODE:
- $H^{+}$ and $Na^{+}$ ions are attracted to the cathode.
- $H^{+}$ is preferentially discharged due to $H^{+}$ being lower in the electrochemical series than $Na^{+}$.
- Each $H^{+}$ ion gains one electron from the cathode to form one hydrogen atom.
- Effervescence of colourless, odourless hydrogen gas is seen at the cathode.
- $2H^{+} (aq) + 2e^{-} \rightarrow H_{2} (g)$
Reaction at ANODE:
- $OH^{-}$ and $Cl^{-}$ ions are attracted to the anode.
- $Cl^{-}$ is preferentially discharged due to the concentration of $Cl^{-}$ being much higher than that of $OH^{-}$.
- Each $Cl^{-}$ ion lose one electron to the anode to form one chlorine atom.
- Effervescence of greenish-yellow chlorine gas is seen at the anode.
- $2Cl^{-} (aq) \rightarrow Cl_{2} (g) + 2e^{-}$
Overall Reaction:
- Hydrogen chloride is decomposed into hydrogen and chlorine gases
- $2HCl (aq) \rightarrow H_{2} (g) + Cl_{2} (g)$
- Volume of chlorine gas collected is slightly less than the volume of hydrogen gas because some chlorine gas dissolves into the electrolyte solution.