Fossil fuels, also known as hydrocarbons, are substances that contain carbon and hydrogen.
- Examples of fossil fuels: Coal, crude oil (petroleum) and natural gas.
- Natural gas consists mainly of methane ($\text{CH}_{4}$).
Complete combustion of fossil fuels (aka burning of fossil fuels) produces carbon dioxide and water.
- It is an exothermic reaction. A large amount of heat energy is released during combustion.
- E.g. Burning methane (natural gas): $\text{CH}_{4} \, \left( \text{g} \right) + 2 \text{O}_{2} \, \left( \text{g} \right) \rightarrow \text{CO}_{2} \, \left( \text{g} \right) + 2 \text{H}_{2}\text{O} \, \left( \text{l} \right) + 882 \, \text{kJ}$
Natural Gas
Natural gas is formed together with petroleum in the ground. Over a period of millions of years, the high pressure and high temperature convert the fossil remains of marine animals and plants into petroleum and natural gas.
$$\text{CH}_{4} \, \left( \text{g} \right) + 2 \text{O}_{2} \, \left( \text{g} \right) \rightarrow \text{CO}_{2} \, \left( \text{g} \right) + 2 \text{H}_{2}\text{O} \, \left( \text{l} \right)$$
$$\Delta \text{H} =-882 \text{kJ mol}^{-1}$$
Methane can also be obtained from coal (via a process – coal gasification).
Coal Gasification
In coal gasification, coal is mixed with oxygen and steam at very high temperature. This results in the breaking of the carbon-carbon bonds in the large hydrocarbon molecules in coal into smaller gaseous hydrocarbons. This process produces syngas (carbon monoxide and hydrogen) and methane gas.
$$\text{C} \, \left( \text{s} \right) + 2 \text{H}_{2} \, \left( \text{g} \right) \rightarrow \text{CH}_{4} \, \left( \text{g} \right)$$
$$\text{C} \, \left( \text{g} \right) + \text{H}_{2}\text{O} \left( \text{g} \right) \rightarrow \text{CO} \left( \text{g} \right) + \text{H}_{2} \left( \text{g} \right)$$
$$\text{C} \, \left( \text{s} \right) + \frac{1}{2} \text{O}_{2} \left( \text{g} \right) \rightarrow \text{CO} \left( \text{g} \right)$$
Syngas can be used directly as a fuel as the burning of carbon monoxide and hydrogen are exothermic processes.
Syngas is a valuable raw material to produce useful chemicals, e.g. methanol.
$$\text{CO} \left( \text{g} \right) + 2 \text{H}_{2} \left( \text{g} \right) \rightarrow \text{CH}_{3}\text{OH} \left( \text{l} \right)$$
Petroleum
Petroleum is a mixture of hydrocarbon molecules – e.g. alkanes, alkenes. The hydrocarbon molecules have different sizes and number of carbon atoms. The smaller molecules have few carbon atoms and low boiling points, while the larger molecules have many carbon atoms and high boiling points. Petroleum is refined to produce useful products and fuels.
Refining of Petroleum
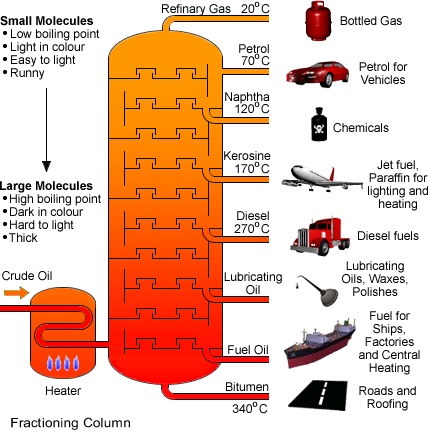
Petroleum can be separated into its useful fractions by fractional distillation in an oil refinery. Each fraction consists of a mixture of hydrocarbon molecules that boils over a range of temperature.
In this process, petroleum is first heated in a furnace. The oil vaporizes and passes up a fractionating column. The fractions comes out of the column at different heights depending on the boiling points. Thus, the refinery gas (petroleum gas) comes out at the top of the column as its molecules have the lowest boiling points. Lubricating oil comes out of the column near the bottom as its molecules have high boiling points. The residue at the bottom of the column is bitumen.
As one goes down the fractionating column, the following changes are observed:
- Their boiling points increase
- The molecules become bigger
- The liquids become more viscous (flow less easily)
- The liquid burn less easily (burns with a more smoky flame)
Uses of fractions of petroleum
| Fraction | Important Uses |
|---|---|
| Petroleum Gas | Bottled gas for cooking |
| Petrol (gasoline) | Fuel for cars |
| Naphtha | Used to manufacture chemicals |
| Kerosene | Jet fuel, fuel for heating and cooking |
| Diesel | Fuel for diesel engines |
| Lubricating oil | Lubricants for machines |
| Bitumen | Used to make road surfaces |