In ionic bonding, the participants are ions and they are kept together by the mutual force of attraction between oppositely charged ions. What are ions?
Ions are charged particles which are formed when an atom loses or gains electrons
- Recall that atoms aim to achieve the stable noble gas configurations (having a fully filled valence shell). Hence, they will try to lose or gain electrons in order to fully fill their valence shell.
- Typically, metal atoms lose their valence electrons to achieve the noble gas configuration. As they lose the valence electrons, they will become positively charged. Hence, we say that a positive ion (cation) is formed.
- Typically non-metal atoms gain valence electrons to achieve the noble gas configuration. As they gain additional electrons, they will become negatively charged. Hence, we say that a negative ion (anion) is formed.
Depending on the position of the metal/non-metal on the periodic table, they will lose/gain different number of electrons. Another way of interpreting this: Depending on the number of valence electrons, they will lose/gain different number of electrons. Some examples to illustrate this:
Consider the element lithium. (metal)
- It has the electron configuration of 2.1.
- The number of valence electron is 1
- It will strive to lose the solo valence electron to end up with an electron configuration of 2.
- The end state is: $\text{Li}^{+}$
Consider the element Aluminium. (metal)
- It has the electron configuration of 2.8.3.
- The number of valence electron is 3
- It will strive to lose the 3 valence electrons to end up with an electron configuration of 2.8.
- The end state is: $\text{Al}^{3+}$
Consider the element oxygen (non-metal)
- It has the electron configuration of 2.6.
- The number of valence electron is 6
- It will strive to gain 2 electrons to end up with an electron configuration of 2.8
- The end state is: $\text{O}^{2-}$
- Note: You might wonder why doesn’t it lose 6 valence electrons. It will have the stable electron configuration of 2 as well. This is because it is more energy efficient to gain 2 electrons than to lose 6 electrons. Hence, the situation of losing 6 valance electrons doesn’t happen.
Consider the element Chlorine (non-metal)
- It has the electron configuration of 2.8.7
- The number of valence electron is 7
- It will strive to gain 1 electron to end up with an electron configuration of 2.8.8
- The end state is: $\text{Cl}^{-}$.
Ionic Bonding
Ionic bonds are formed between metal atoms and non-metal atoms. During the formation of ionic bonds, valence electrons from the metal atoms are transferred to the non-metal atoms. This result in the formation of positively-charged ions (cations) and negatively-charged ions (anions). Hence, there will be strong electrostatic forces of attraction between the oppositely charged ions which is termed as ionic bonding.
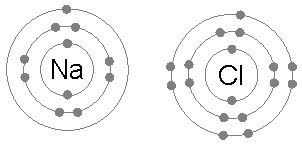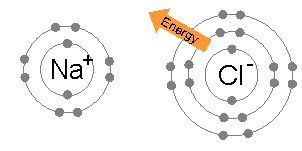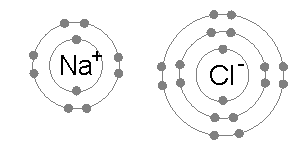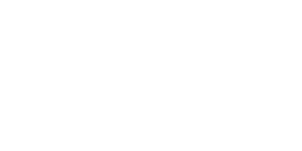
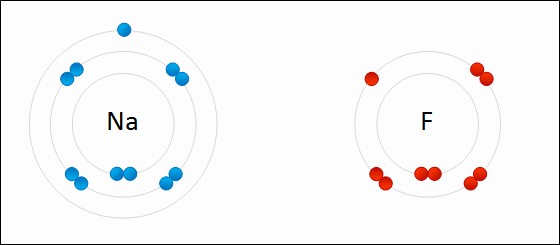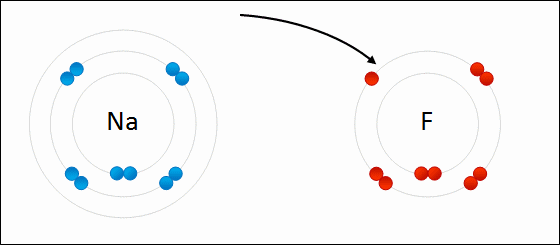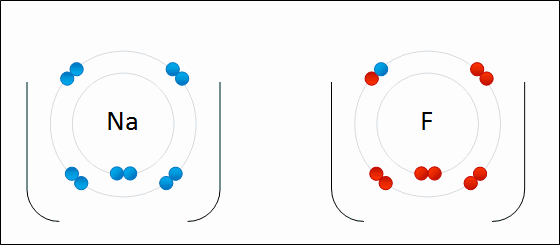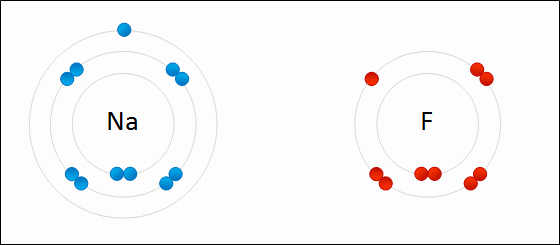
Sodium chloride as an example of ionic bonding
- Recall that sodium has electron configuration of 2.8.1 and chlorine has an electron configuration of 2.8.7
- The sodium will want to lose 1 electron while chlorine will want to gain 1 electron
- Hence, the sodium will transfer the valence electron to chlorine
- This will result in $\text{Na}^{+}$ and $\text{Cl}^{-}$
- The oppositely charged ions will then be held together by strong electrostatic forces of attraction to form an ionic bond.
Another animation of ionic bonding: (Featuring NaF)
Properties of ionic compounds
- Ionic compounds have high melting and boiling points
- Ionic compounds do not conduct electricity in the solid state.
- Ionic compounds conduct electricity when molten or dissolved in water
- Ionic compounds readily dissolve in water but do not dissolve in organic solvent
Reason for high melting and boiling point: The oppositely charged ions are held together by very strong electrostatic forces of attraction which require a lot of energy to overcome. The ability to withstand high temperature makes ionic compounds suitable for use as lining for furnaces.
Reason for electrical conductivity: In the solid state, the ions are held together by very strong electrostatic forces of attraction and can only vibrate about a fixed point. Hence, the ions cannot move freely to conduct electricity. However, in the molten or aqueous (dissolved in water) state, the ions are free to move around (= conduct electricity).