Recall: Types of states of matter: Solid, Liquid and gas.
Matter can change from one state to the other. The change in the state is
- Reversible
- A result of changes in pressure or temperature (usually in the form of heat absorbed or released)
Melting
Melting is the process whereby a solid changes into a liquid.
- The temperature at which melting occurs is called the melting point.
- Melting point is affected by pressure. One example is water. The melting point of water is lowered with increased pressure. Note: Water is special. Most of the other substances will have a higher melting point with higher pressure.
- Heat is absorbed during melting.
- Impurities in the solid will lower the melting point of the solid.
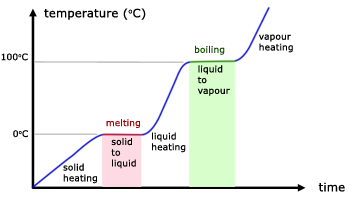
From the graph above,
- $0^{\circ}$C is the melting point of the solid.
- Temperature remains constant at $0^{\circ}$C as the solid is melting.
- During the melting process, solid and liquid exist in equilibrium.
Step by step process of what happens during melting:
- Heat energy is absorbed by the particles
- Heat energy is converted to kinetic energy
- The kinetic energy of the particles increases and the particles in the solid vibrate faster
- At melting point, the particles have gained enough energy to overcome the attractive forces between particles
- Particles starts to move away from their fixed position
- Liquid is formed
Cause for constant temperature during melting: The absorbed heat energy during melting is used to weaken the attractive forces between particles and not the kinetic energy of the particles.
Boiling
Boiling is the process whereby a liquid changes into solid.
- Similar to melting, heat is absorbed during boiling.
- The temperature at which boiling occurs is called the boiling point.
- Boiling point is affected by pressure. Typically, higher pressure will result in higher boiling point.
- A pure liquid has a fixed boiling point.
- Impurities will affect the boiling point of the liquid
From the graph above,
- $100^{\circ}$C is the boiling point of the liquid
- Temperature remains constant at $100^{\circ}$C as the liquid is boiling
- During the boiling process, liquid and gas exist in equilibrium.
Step by step of what happens during boiling:
- Heat energy is absorbed by the particles
- The kinetic energy of the particles increases
- At boiling point, the particles have enough energy to overcome the attractive forces between the particles and move away from one another.
- The particles escape from the liquid to form gas
Cause for constant temperature during boiling: The absorbed heat energy is used to overcome the attractive forces between the particles and not the kinetic energy.
Evaporation
Evaporation is the process whereby a liquid changes into gas (similar to boiling but different)
- The change in state from liquid to gas only takes place at the surface of the liquid
- Heat is absorbed during evaporation
- There is no fixed temperature at which evaporation takes place. Evaporation takes place at all temperature below the boiling point of the liquid.
Condensation
Condensation is the process whereby a gas changes to a liquid.
- The change in state takes place when the gas is cooled to its boiling point.
- Heat is released to the surroundings during condensation.
Freezing
Freezing is the process whereby a liquid changes into a solid
- The change in state takes place when the liquid is cooled to its freezing point.
- Impurities will affect the freezing point of the substance.
Sublimation
Sublimation is the process whereby a solid changes into a gas without going through the liquid phase.
- Heat is absorbed to change from solid to gas.
- Examples of solid that undergoes sublimation: Dry ice, ammonium chloride.
Can you pls add the states of matter like solid liquid and gas with diagrams and their differences for higher classes for better understanding and by the way I love your website it is very helpful