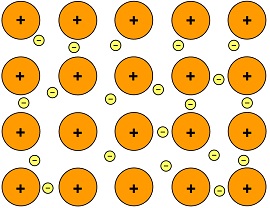
Metals have a tendency to lose their valence electrons in order to achieve the stable noble gas configuration. We say that metals are electropositive (tendency to lose electrons).
Step by step guide to metallic bonding
- Atoms in metals has valence electrons.
- The atoms pool all their valence electrons to form a “sea of electrons”. The metal atoms are now positive ions due to the loss of electrons.
- These electrons move freely among the positive metal ions
- Metallic bonding is the electrostatic force of attraction between the electrons and the positive ions
Hence, a metal is a giant structure consisting of regularly arranged and closely packed positive metal ions in a ‘sea of electrons’. The metal ions are big and cannot move, and the electrons are very small and can move freely throughout the metal.
Properties of metals:
- High melting and boiling points $\rightarrow$ Due to the strong electrostatic force of attraction between the electrons and positive ions. (Note: Mercury is an exception as it has a low melting point)
- Conduct electricity in solid and molten state $\rightarrow$ Have free electrons to conduct electricity
- Conduct heat $\rightarrow$ Have free electrons to conduct heat
- Malleable (can be bend) $\rightarrow$ Due to the orderly packing of metal ions. The layers of metal ions can slide easily over one another when a force is applied.
- Ductile (can be stretched without breaking)
How to strengthen metal?
By making it into an alloy. An alloy is a mixture of metallic elements. Some may contain non-metallic elements. Alloys are stronger and harder than pure metals. This is because in alloys, the metal ions are of different sizes and this disrupts the orderly layers of metallic ions in a metal and makes it much harder for the layers to slide over each other, hence making it harder, stronger, less malleable and ductile.