Preparation of Soluble Salts
The methods of preparing soluble salts are categorised into:
- Solution + Solid
- Solution + Solution
Solution + Solid
In the “solution + solid” method, a solid and solution are reacted together to form soluble salt.
You may follow the following procedures to prepare soluble salts:
- Add solid, a little at a time into warm solution. Stir.
- Filter away the excess solid.
- Evaporate the filtrate.
- Depending on the heat stability of the salt, you can either dry the salt with heat or crystallise the salt.
Note: The procedure for crystallising are as follows:
- Heat the solution gently until a thin film of crystals is formed on the surface.
- Cool saturated solution slowly.
- Filter out the crystals.
- Dry crystals between sheets of filter papers.
Note 2: Excess solid reactants must be used. This is because it is easy to separate the excess solids from the resulting salt solution by filtration. If the solution is in excess, it will contaminate the salt solution as it cannot be easily separated from the salt solution.
Reacting Acid + Metal (excess)
Examples of such reactions:
$$\text{Zn}(\text{s}) + \text{H}_{2}\text{SO}_{4}(\text{aq}) \rightarrow \text{ZnSO}_{4}(\text{aq}) + \text{H}_{2}(\text{g})$$
$$\text{Fe}(\text{s}) + 2 \text{HCl}(\text{aq}) \rightarrow \text{FeCl}_{2}(\text{aq}) + \text{H}_{2}(\text{g})$$
You may observe that the solid metal will dissolve slowly in the acid and that effervescence of colourless, odourless gas is seen.
How to tell that the reaction has stopped: No more effervescence is observed.
Notes:
- There are several metals that cannot be used:
- $\text{K}$, $\text{Na}$: They are too reactive. They will react explosively with acid.
- $\text{Pb}$, $\text{Cu}$, $\text{Hg}$, $\text{Ag}$, $\text{Au}$: They are too unreactive. They do not react with acids.
- The acid is typically heated up to speed up the reaction.
Reacting Acid + Carbonate (insoluble, excess)
Examples of such reactions:
$$\text{CaCO}_{3}(\text{s}) + 2 \text{HCl}(\text{aq}) \rightarrow \text{CaCl}_{2}(\text{aq}) + \text{CO}_{2}(\text{g}) + \text{H}_{2}\text{O}(\text{l})$$
$$\text{CuCO}_{3}(\text{s}) + \text{H}_{2}\text{SO}_{4}(\text{aq}) \rightarrow \text{CuSO}_{4}(\text{aq}) + \text{CO}_{2}(\text{g}) + \text{H}_{2}\text{O}(\text{l})$$
How to tell that the reaction has stopped: No more effervescence is observed.
Reacting Acid + Base (insoluble, excess)
Examples of such reactions:
$$\text{PbO}(\text{s}) + 2\text{HNO}_{3} \rightarrow \text{Pb}(\text{NO}_{3})_{2}(\text{aq}) + \text{H}_{2}\text{O}(\text{l})$$
$$\text{CuO}(\text{s}) + \text{H}_{2}\text{SO}_{4}(\text{aq}) \rightarrow \text{CuSO}_{4}(\text{aq}) + \text{H}_{2}\text{O}(\text{l})$$
$$\text{Pb}(\text{OH})_{2} + 2\text{HNO}_{3} \rightarrow \text{Pb}(\text{NO}_{3})_{2}(\text{aq}) + 2 \text{H}_{2}\text{O}(\text{l})$$
How to tell that the reaction has stopped: The solid reactant no longer dissolve in the acid.
Solution + Solution
In the “solution + solution” method, a solution and another solution are reacted together to form soluble salt.
The reactants must all be used up so that the reaction mixture only contains the required salt and other “harmless” products. Hence, exact quantities of the reactants must be used to ensure complete reaction. A method that allows this is titration.
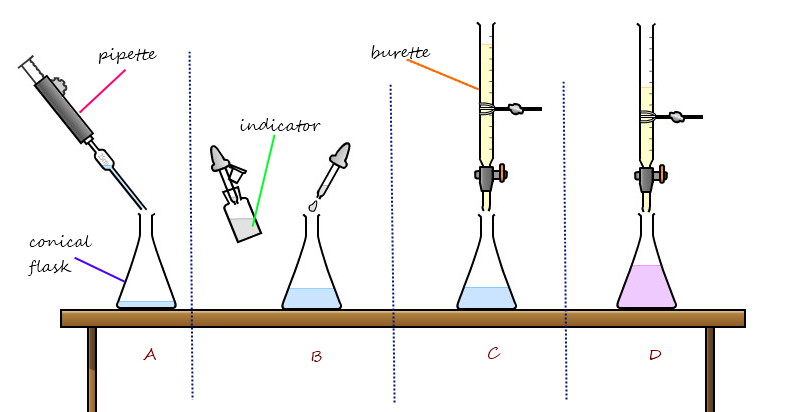
You may follow the following titration procedures to prepare soluble salts:
- Pipette a volume of acid into a conical flask.
- Add a few drops of indicator into the conical flask.
- Using a burette, add the alkali, a little at a time, into the conical flask until the indicator changes colour.
- Note the volume of alkali used and throw away the solution in the flask.
- Repeat the titration without the indicator, using the same volume of alkali and a clean flask.
- Evaporate the mixture obtained.
Reacting Acid + Alkali
Examples of such reactions:
$$\text{H}_{2}\text{SO}_{4}(\text{aq}) + 2\text{NaOH}(\text{aq}) \rightarrow \text{Na}_{2}\text{SO}_{4}(\text{aq}) + 2 \text{H}_{2}\text{O} (\text{l})$$
$$\text{NH}_{3} + \text{H}_{2}\text{O} (\text{l}) + \text{HCl}(\text{aq}) \rightarrow \text{NH}_{4}\text{Cl}(\text{aq}) + \text{H}_{2}\text{O}(\text{l})$$
How to tell that the reaction has stopped: The solution turns orange (as the indicator used is methyl orange).
Reacting Acid + Carbonate (aqueous)
$$\text{K}_{2}\text{CO}_{3}(\text{aq}) + 2 \text{HNO}_{3}(\text{aq}) \rightarrow 2 \text{KNO}_{3}(\text{aq}) + \text{H}_{2}\text{O} (\text{l}) + \text{CO}_{2} (\text{g})$$
$$(\text{NH}_{4})_{2}\text{CO}_{3}(\text{aq}) + \text{H}_{2}\text{SO}_{4}(\text{aq}) \rightarrow (\text{NH}_{4})_{2}\text{SO}_{4}(\text{aq}) + \text{H}_{2}\text{O}(\text{l}) + \text{CO}_{2}(\text{g})$$
How to tell that the reaction has stopped: No more effervescence is observed.
Preparation of Insoluble Salts
Precipitation (solution + solution)
Insoluble salts are prepared by precipitation via a double decomposition method, as shown below:
$$\text{M}_{1}\text{A}(\text{aq}) + \text{M}_{2}\text{B}(\text{aq}) \rightarrow \text{M}_{1}\text{B}(\text{aq}) + \text{M}_{2}\text{A}(\text{aq})$$
Notice that A and B are swapped.
You may follow the following procedures to prepare insoluble salts:
- Mix the solutions.
- Filter out the precipitate.
- Wash residue with deionised water.
- Dry precipitate between sheets of filter paper.
Solube alkalis dissolve in water to form solube alkaline hydroxides like Na,K &NH4 only but others dont dissolve but for insolube alkaline hydroxides in form of precipates
Best site ever. In practicles on salt preparation i got an “A” just becoz of this site
What about revision questions
what the different btn soluble and insoluble alkalis?
I always like to study more about from this site because, of clear writing that are easy to read and observe , correct explaination are also there.from kidega Emmanuel from gulu town , uganda.i wish you well.
Good and clear what next can I said ,is a quiet side for reading,and it’s a very useful.